by Sarah Diregger
While cooking, one can notice that metal becomes hot very quickly and wood or plastic is safer to touch if you’re not in the mood for a 2nd-degree burn. I’m sure everybody knows not to touch the pan while it sits on the stove. But why? What’s the difference between metal and plastic? Why does metal heat up so easily?
Firstly, to prove my theory that metal heats up faster than plastic I experimented:
You need a plastic rod, a metal rod, an infrared camera, and warm hands. You hold each rod in your hands for 1-2 minutes. Then you point the infrared camera at the two rods, and you can see the results immediately.
Here, you can see an image of the two rods next to each other before the experiment: It’s very faint but I think you can make out two different sticks. The left one is metal, the right one plastic.
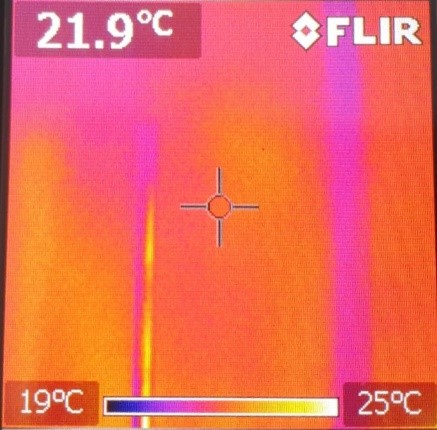
This is what the plastic rod looks like after one hand held it:

This is what the plastic rod looks like after 3 hands held it:

This is what the metal rod looks like after a you hold it in your hand:
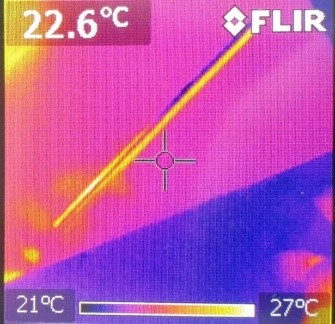
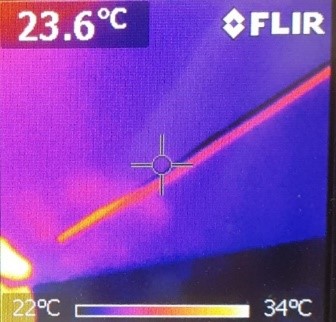
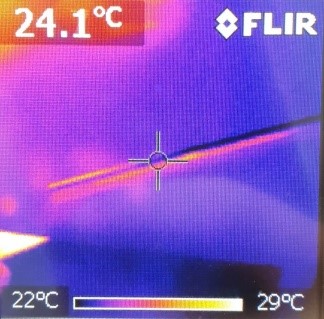
On the scale at the bottom of the images, you can see which color symbolizes which temperature. Consequently, we can see that before the experiment, both were at about the same temperature. After the experiment, the plastic rod was only heated at the area you held it. However, the temperature of the warm hands spread farther and the area of warmth was greater than the size of your hands.
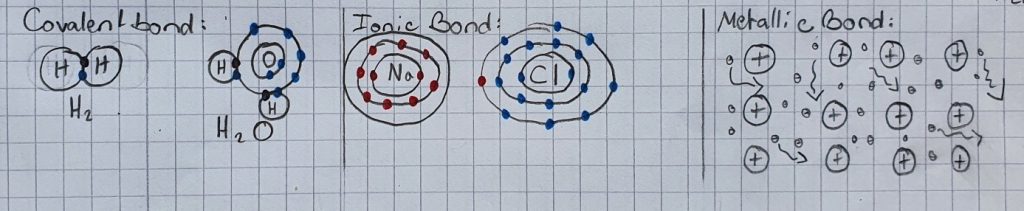
The explanation for this phenomenon lies on the atomic level. It’s important to know that atoms form 3 different types of bonds:
Covalent bond: Bond, in which atoms share electrons
Ionic bond: One atom gives its valence (outermost) electron(s) to the other atom. Therefore, one atom acquires a positive charge and the second acquires a negative charge. Positive and negative attract each other, leading to the atoms forming a bond.
Metallic bond: The nuclei (plural of nucleus) of the atoms arrange themselves in a fixed structure, while the negatively charged electrons move freely in between the positively charged nuclei.
Here is an illustration to help you visualize what this looks like:
Before I explain further, it’s crucial to know what heat is. Heat is basically the vibration of atoms and molecules within a substance. The more they vibrate, the hotter the substance is.
We will be focusing on the last type of bond, the metallic bond. Since the electrons don’t have a fixed position within the atomic structure, they can move around more than the electrons in a covalent or ionic bond. If we look at the physical properties of heat, it’s obvious why metals, who have metallic bonds, conduct heat better. The electrons can vibrate easier and it’s easier for them to pass this vibration on to the next electrons. Plastic has a covalent bond. Therefore, the atoms within the polymer (the scientific name for plastic) are tied together tighter and vibrations can’t be transmitted as easily.
In conclusion, metals transfer heat the best because of the type of atomic bonds they have. The metallic bond allows electrons to move between the nuclei. Therefore, the movement of heated electrons can be conveyed easier than in fixed bonds. Which important lesson do we learn in our day-to-day lives? Never use metal if you want to handle something hot because it doesn’t shield the heat.
Sources:
https://www.edinformatics.com/math_science/why_metals_conduct.htm#:~:text=Metal%20is%20a%20good%20conduction,of%20their%20energy%20to%20them.
https://www.britannica.com/science/crystal/Conductivity-of-metals
https://www.lernhelfer.de/schuelerlexikon/chemie/artikel/waermeleitung#
https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Chemical_Bonding/Fundamentals_of_Chemical_Bonding/Metallic_Bonding
https://www.chemie.de/lexikon/Metallische_Bindung.html#:~:text=Als%20Metallische%20Bindung%20oder%20Metallbindung,Metallen%20und%20in%20Legierungen%20vorliegt.&text=Sie%20wird%20durch%20Anziehungskr%C3%A4fte%20zwischen%20Metall%2DIonen%20und%20freien%20Elektronen%20verursacht.
https://www.lernhelfer.de/schuelerlexikon/chemie/artikel/metallbindung
[last access: 24.01.2022]