by Michael Himmelbauer
A diver inside a water bottle sinks to the ground with the help of magic powers of your fingers. What sounds to be fake, can easily be rebuilt by using some materials you may have at home, but can also be found in our school’s Physics cabinet:
- a water bottle with a volume of approximately 1.5 to 2 liters (as physicists, we say 0.0015 to 0.002 m3 [one cubic meter consists of 1000 cubic decimeters and one liter has the same volume as one cubic decimeter])
- a tiny plastic tube that has a hole in one transversal side
- a cork that has almost the same diameter as the plastic tube
- and of course: Dihydrogen monoxide (H2O), also known as water
At first, you need to insert the cork into the tube and the tube into the water bottle as shown in the picture below (therefore, it is crucial that there is air above the cork inside the tube and the „diver“ is at the top of the bottle):

When pressing your hands onto the bottle, you might be surprised by seeing the diver gliding to the bottom of the bottle, the stronger you press the wider it glides. As soon as you stop pushing, it ascends back to the top.

But why does that occur? Do your fingers have magic powers that make a diver glide inside a water bottle? Or is Dihydrogen monoxide an unusual liquid?
As a first explanation, it can be mentioned that the water inside the bottle can be replaced by another liquid as well, that means it isn’t a special substance (at least not concerning this experiment). So don’t worry, you can go on drinking it.
Before explaining this phenomenon, we need to take a look at a physical force called buoyancy. What the hell is that? It’s the force that makes a balloon filled with the gas Helium (chemical symbol in the periodic table of elements: He) ascend and an air-filled ball (air that surrounds us consists of nitrogen (N2), oxygen (O2), carbon dioxide (CO2) and some other low-concentrated gases) rise to the top of a water-filled swimming pool when pushing it to the ground.The buoyancy force has got the same value as the weight force of the displaced water which can be calculated easily by using the following formula:
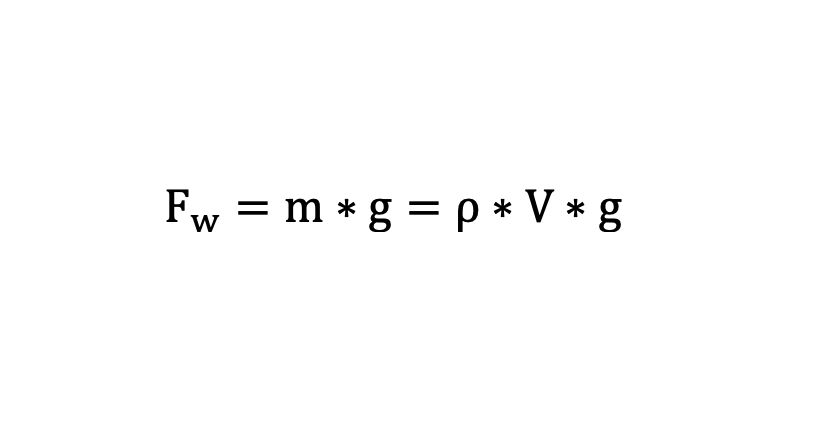
[Fw = weight force, m = mass, g = gravitational acceleration (= 9.81 m/s2), ρ = density of water (= 1000 kg/m3), V = volume]
When investigating the formula, we can derivate that when the volume rises, there is a higher weight force and consequently also a „stronger“ buoyancy force. Invertedly, a drop in volume means a lower weight force and a „weaker“ buoyancy force.
Keeping these facts in mind, we can examine the effects in the experiment:
Due to the force of your fingers, the pressure inside the water bottle rises and that is why the cork is pressed into the tube. As a result, the tube’s volume decreases and as there is a direct proportionality between volume and buoyancy force, it declines as well. This can be recognized as the „diver“ (which still has got the same weight as the air in the tube is merely compressed) sinks to the bottom of the water bottle.
But don’t worry, you haven’t damaged anything: As soon as you stop pressing the bottle with your fingers, the tube ascends to the top again, so it’s reversible.
For illustration, I made a drawing to convey the theory:
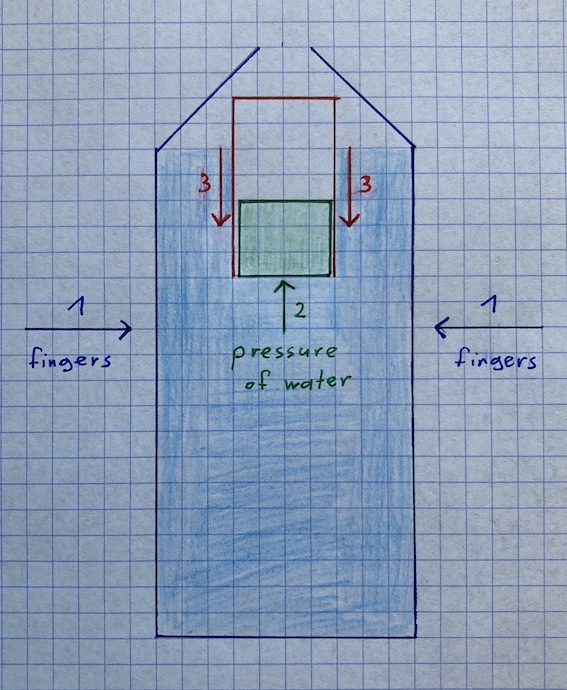
Now you might ask yourself: Are there also some practical applications of buoyancy that are maybe easier than the orthography of the term?
Fortunately, there aren’t only some, but many, rather countless:
As mentioned above, buoyancy is commercially used for balloon flights as it makes the balloon rise into the sky. This can be explained by outlining that Helium has a lower density than air (that means that a specific volume of Helium is lighter than the same volume of air) and that is why it can ascend and even compensate the weight of the basket that carries the people and gives them the opportunity to enjoy the weather and be amazed by the landscape.
Another application of that magic, difficultly pronounceable force are life jackets that must be provided on every boat and ship as well as in airplanes. As the material has a way lower average density than water, it can even prevent an adult from drowning when the mode of transportation has an accident.
To sum up, it can be stated that buoyancy force is hard to write and pronounce, but crucial for our everyday life. It can be used for impressing spectators by convincing them that you’ve got magic powers to make a cork diver sinking – although you just press your fingers onto a common bottle filled with usual water.
source:
Putz, Bruno: Faszination Physik 1+2. Linz: Veritas 2011, p. 160-165
fotocredit: © by Michael Himmelbauer

